Image 1 of 4
Image 1 of 4

 Image 2 of 4
Image 2 of 4

 Image 3 of 4
Image 3 of 4

 Image 4 of 4
Image 4 of 4





AP Chemistry Endothermic Exothermic Processes Spin and Cover Partner Activity
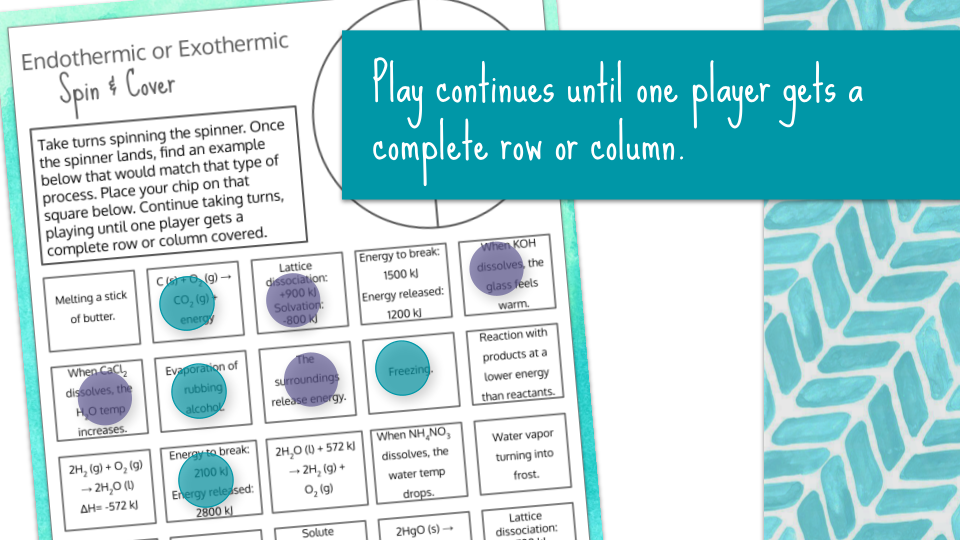
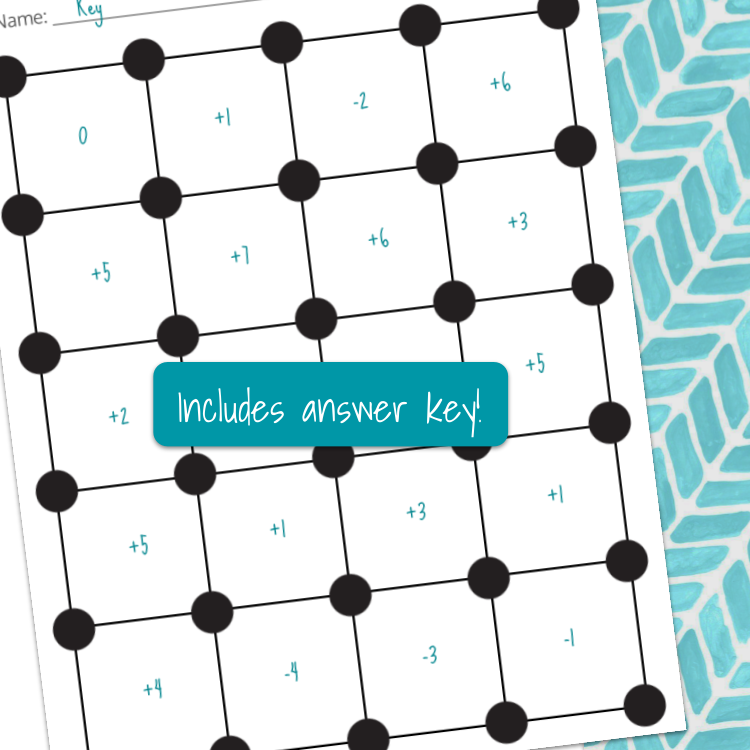
This partner activity makes for great practice identifying processes as endothermic or exothermic! Students will spin a wheel (spinner not included), which will provide them with endothermic or exothermic. Students will then locate an appropriate process in the grid that fits in that category.
For example, if the spinner lands on endothermic, then the student would locate a process in the grid that is an endothermic process. The player will then place a marker/chip (not included) on an appropriate square. Students will take turns spinning the wheel and placing chips until one player completes a row or column.
This spin and cover activity includes:
phase changes
reactions with energy included on reactant or product side
reactions with a ΔH value included (positive or negative)
energy required to break bond(s) and energy required released when forming bonds
change in temperature of water when a solute is dissolved
This resource comes in a printable pdf, in both color and an ink-saving version. Each mat will need a spinner and markers/bingo chips, which are not included.
Join my email list and receive four self-checking ideas, along with four FREE self-checking products and/or templates.
Let's connect on Instagram.
This partner activity makes for great practice identifying processes as endothermic or exothermic! Students will spin a wheel (spinner not included), which will provide them with endothermic or exothermic. Students will then locate an appropriate process in the grid that fits in that category.
For example, if the spinner lands on endothermic, then the student would locate a process in the grid that is an endothermic process. The player will then place a marker/chip (not included) on an appropriate square. Students will take turns spinning the wheel and placing chips until one player completes a row or column.
This spin and cover activity includes:
phase changes
reactions with energy included on reactant or product side
reactions with a ΔH value included (positive or negative)
energy required to break bond(s) and energy required released when forming bonds
change in temperature of water when a solute is dissolved
This resource comes in a printable pdf, in both color and an ink-saving version. Each mat will need a spinner and markers/bingo chips, which are not included.
Join my email list and receive four self-checking ideas, along with four FREE self-checking products and/or templates.
Let's connect on Instagram.